BC Platforms signs Strategic Collaboration Agreement with AWS to accelerate healthcare and life sciences innovation
BC Platforms signs a Strategic Collaboration Agreement with AWS to scale secure, AI-powered research environments.

Easily explore complex genomics, clinical data, and other rich information using an intuitive AI-powered platform that supports the entire drug lifecycle.
BC Catalyst is built on a powerful, proprietary AI-driven foundation that enriches our datasets with public-domain intelligence, delivering a 360° view of the precision medicine landscape. Featuring a pharma-grade UX, BC Catalyst makes actionable insights easy to access and enables fast, confident decision-making.
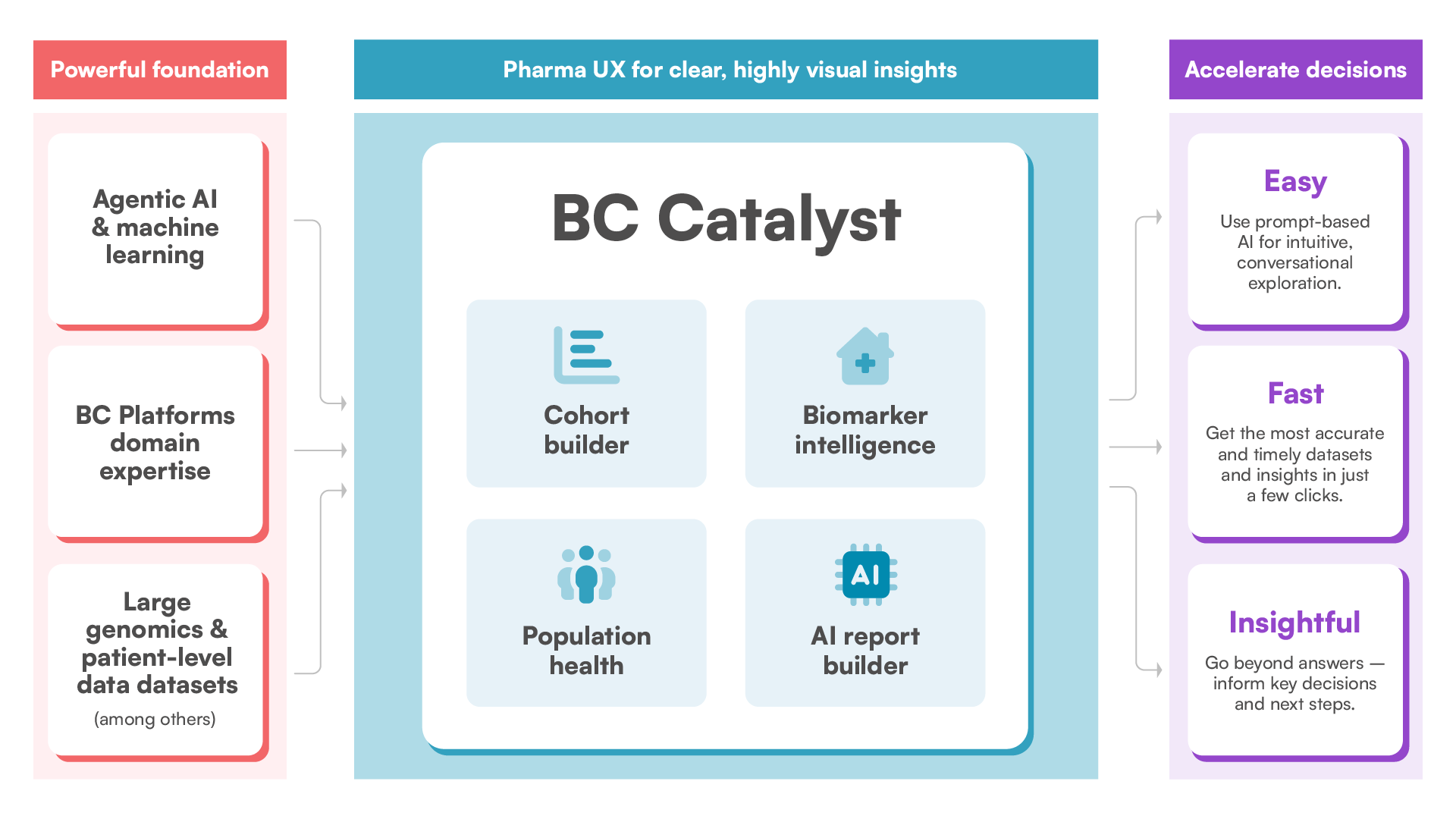
Accelerate your research with BC Catalyst – an intuitive SaaS-based platform featuring prompt-based cohort building, biomarker discovery, and standard of care insights. By combining genomic, clinical, real-world data, and inputs from other public sources in an agentic AI model that continues to learn and grow, BC Catalyst fuels precision medicine strategy, market access, and evidence generation.
Quickly build and query patient cohorts using harmonized genomic and clinical data. Define, filter, and segment populations across disease areas to support research, trial planning, and commercialization strategies.
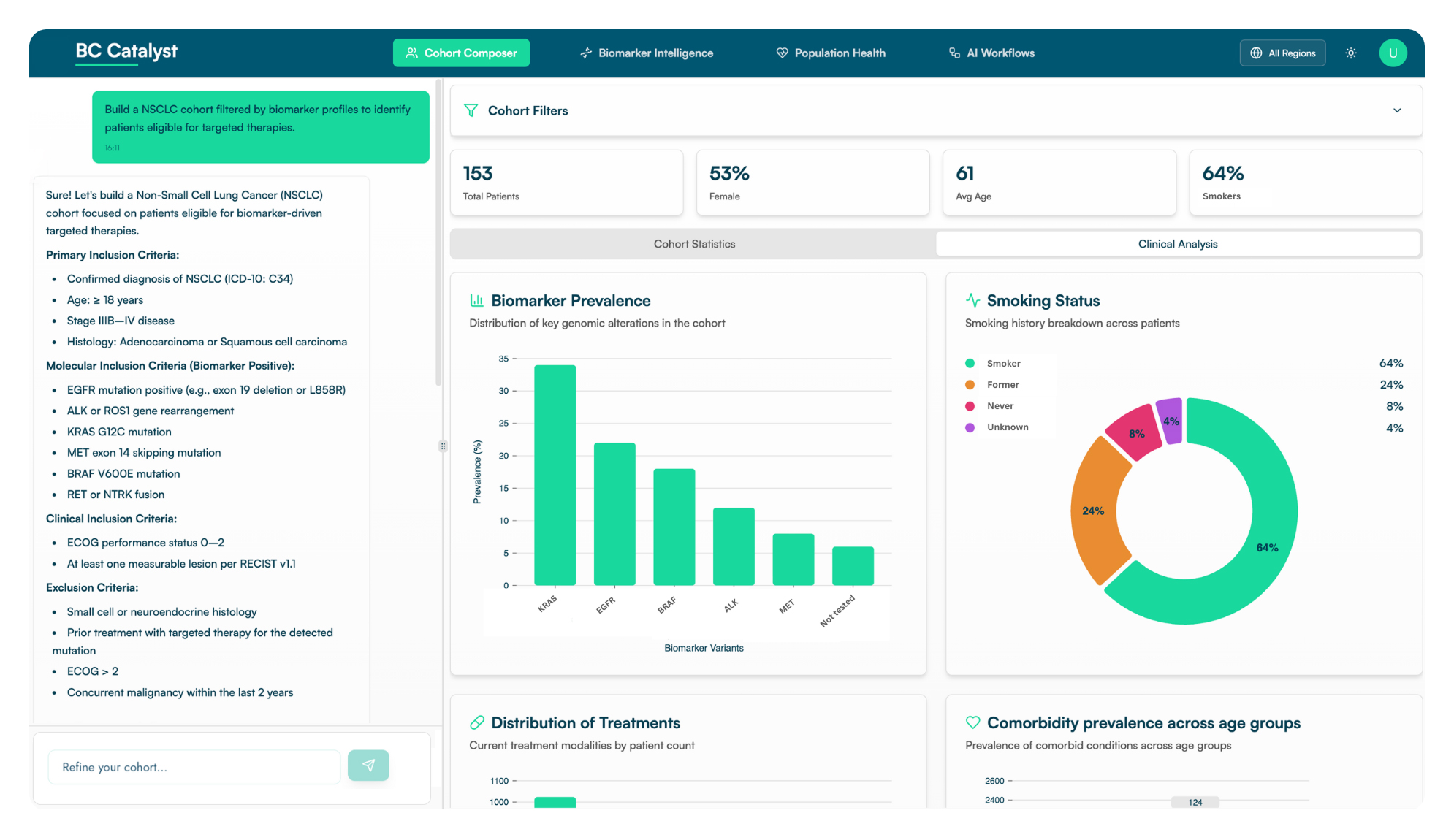
Unlock actionable insights into biomarker-drug-response relationships. Accelerate drug discovery, optimize therapy selection, and predict late-stage failures with advanced analytics and visualizations in just a few clicks.
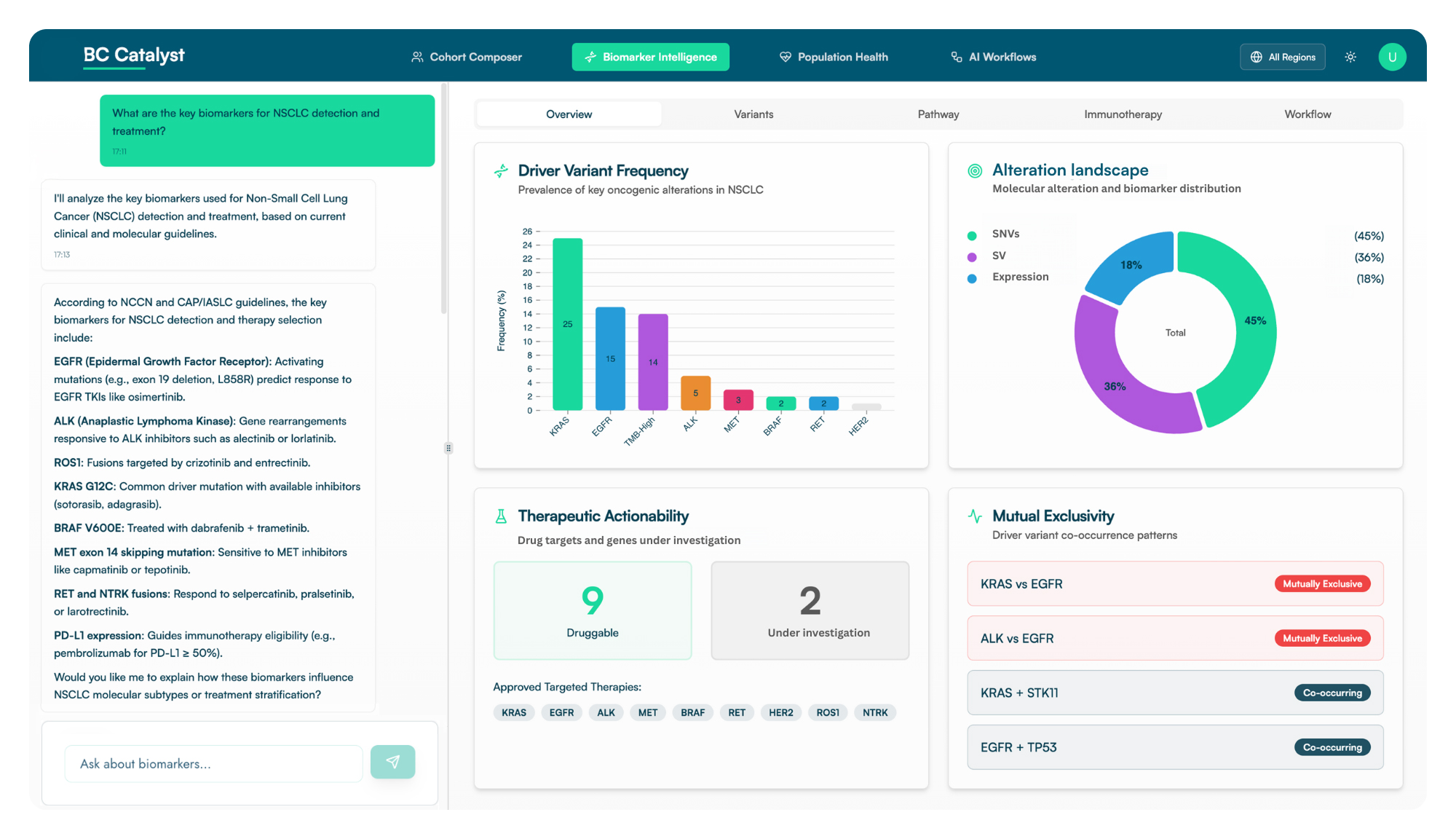
Reveal population-level health trends, real-world outcomes, and evolving standards of care. Visualize patient journeys, treatment patterns, and market opportunities to inform value-based care and commercial strategies that position your product for success.
Create tailored reports by pulling information from all modules in BC Catalyst. AI-supported workflows and visualizations make it easy to share insights across teams – all in a few clicks and in a model that’s continually learning as you use it.


BC Catalyst is powered by BC Unify, which acts as the integration engine behind our solutions — ingesting, enriching, and transforming diverse data into a harmonized structure that supports advanced analytics and healthcare research.
Experience the future of data exploration with BC Catalyst’s agentic AI – prompt-based intelligence and pharma-grade UX that make complex analysis intuitive for every user.
Enables intuitive, conversational data exploration for all users through an agentic AI model built specifically for life sciences by our domain experts.

Integrates data, compliance, and usability for translational pharma teams to save time, provide peace of mind, and simplify initial analysis.

Creates an array of data visualizations such as bar charts, pie charts, Sankey diagrams, and risk maps for easy sharing and further exploration.
Supports rapid adoption within and across teams through a web-based platform that’s available via subscription – no installation required.
Reduce time-to-market through optimized target identification and patient stratification.
Increase treatment success rates through precision patient matching.
Minimize late-stage failures and safety concerns through predictive analytics.
Establish a competitive advantage in the rapidly expanding precision medicine market.
BC Catalyst delivers significant value to teams across your entire organization – from early-stage discovery through clinical development, product launch and beyond.
BC Platforms meets the highest international standards for data protection, product quality, and ethical data governance. Our ISO-certified systems and compliance frameworks protect every stage of the data lifecycle, ensuring partners can innovate confidently and responsibly.
BC Platforms signs a Strategic Collaboration Agreement with AWS to scale secure, AI-powered research environments.
A closer look at how RWD, analytics, and AI are reshaping standard of care insights and evidence generation across the drug lifecycle.
BC Platforms earns the SEF.Growth High‑Potential label for secure, scalable innovation in health data.